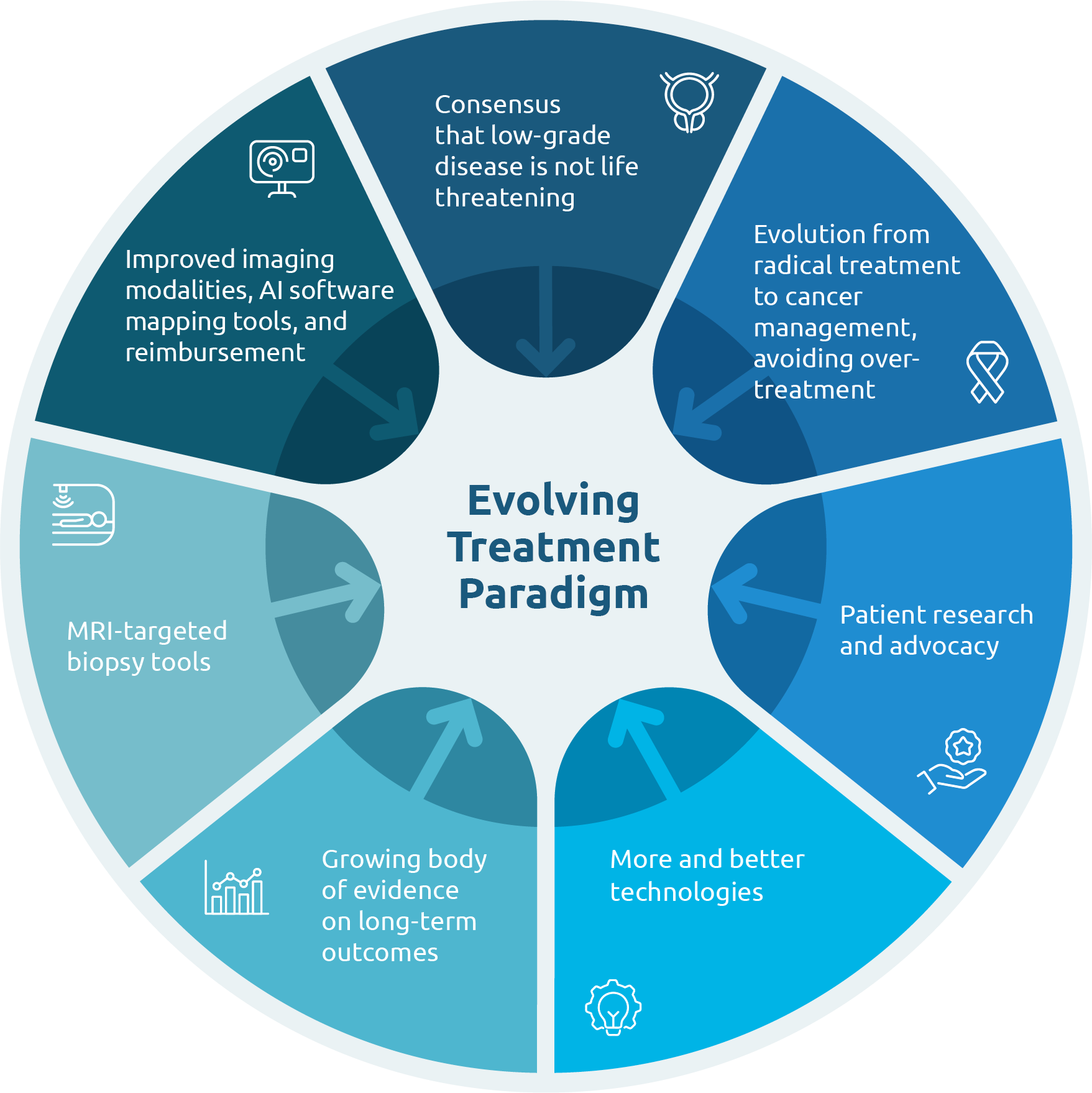
Clinical Evidence.
VAPOR 2 (n=110)
Clinical Data and Patient Satisfaction Results:
91%
clearance of targeted MRI visible ≥GG2 disease following a single treatment
2.7%
≥ Grade 2 urinary incontinence
No device related
serious adverse events
Low rates of
erectile dysfunction
93%
of patients were extremely satisfied or satisfied with the Vanquish procedure at 12 months (n=107)
94%
of the patients believed that the Vanquish procedure was a good decision (n=107)
Minimal to no pain
Most patient reported no pain.



 Refer to the device User Manual for a list of contraindications, warnings, and cautions.
Refer to the device User Manual for a list of contraindications, warnings, and cautions. Training: Do not operate the Vanquish System without completing Francis Medical-provided physician training. Untrained operation of the device may lead to improper use. Improper use can result in patient injury or equipment malfunction.
Training: Do not operate the Vanquish System without completing Francis Medical-provided physician training. Untrained operation of the device may lead to improper use. Improper use can result in patient injury or equipment malfunction.