[ditty id=4186]
Stay
Up-To-Date
Articles, publications, podcasts, and more to keep you current on our Vanquish Water Vapor Ablation System.

Francis Medical Announces First Commercial Vanquish® Procedure to Ablate Prostate Tissue in the Midwest
Minneapolis [April 21, 2026] – Francis Medical, Inc., a privately held medical device company based out of Maple Grove, MN, today announced that Aaron Milbank, MD. of Minnesota Urology has successfully completed the first commercial procedure in the Midwest using the Vanquish® Water Vapor Ablation System, a minimally invasive technology designed to ablate targeted prostate tissue....
Learn More >

Francis Medical Announces Further Commercial Expansion of Vanquish® Water Vapor Ablation with First Commercial Procedures to Ablate Targeted Prostate Tissue in the Southeast
Minneapolis, MN [April 16, 2026] – Francis Medical, Inc., a privately held medical device company, today announced the continued expansion of its U.S. commercial launch with the first commercial procedures performed using the Vanquish® Water Vapor Ablation System in the Southeast, a minimally invasive technology designed to ablate targeted prostate tissue. ...
Learn More >

Francis Medical Announces First Commercial Vanquish® Procedure on the East Coast for Prostate Tissue Ablation
Minneapolis, MN [March. 25, 2026] – Francis Medical, Inc., a privately held medical device company, announced today the successful completion of the first commercial (non-clinical study) procedure on the East Coast using the Vanquish® Water Vapor Ablation System, a minimally invasive technology designed to ablate targeted prostate tissue while potentially helping to preserve urinary and sexual function....
Learn More >

Francis Medical Announces First Commercial Vanquish® Procedures in Arizona for Prostate Tissue Ablation
MINNEAPOLIS (March 5, 2026) – Francis Medical, Inc., a privately held medical device company, announced today the successful completion of the first commercial (non-clinical trial) procedures in Arizona using the Vanquish® Water Vapor Ablation System for prostate tissue ablation....
Learn More >
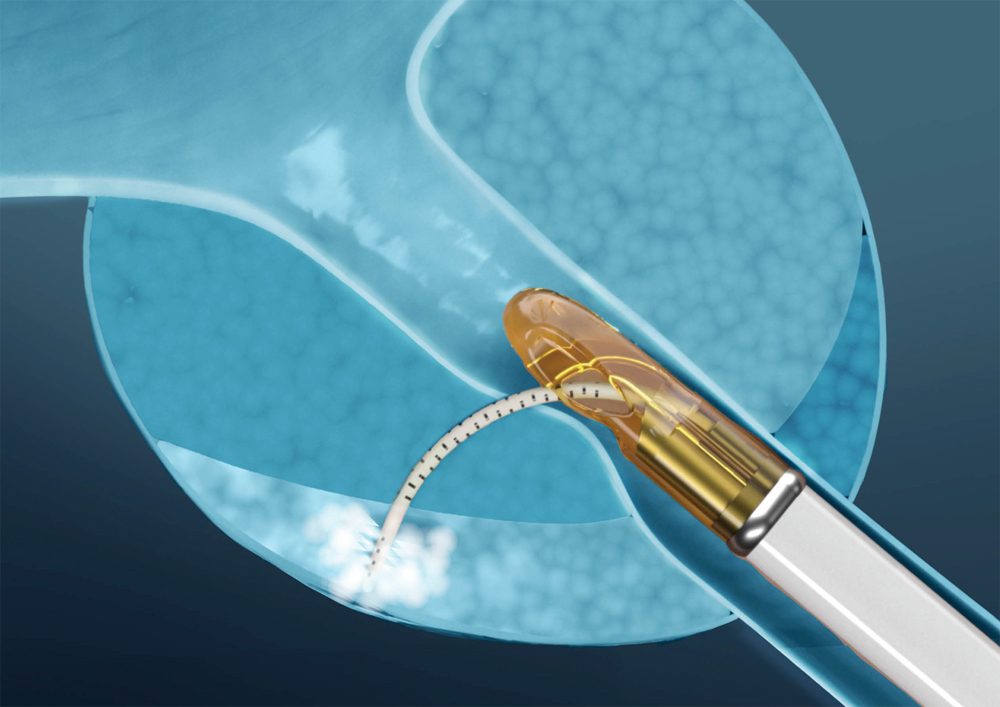
Francis Medical Announces First Commercial Procedure with the Vanquish® Water Vapor System for Prostate Tissue Ablation
MINNEAPOLIS (Feb 3, 2026) – Francis Medical, Inc., a privately-held medical device company, announced the first commercial procedure using the Vanquish Water Vapor Ablation System...
Learn More >

Francis Medical Receives FDA 510(k) Clearance for Use of the Vanquish® Water Vapor System for Prostate Tissue Ablation
MINNEAPOLIS (Dec. 2, 2025) – Francis Medical, Inc.,a privately-held medical device company developing the breakthrough Vanquish® Water Vapor Ablation System ...
Learn More >
Follow Us
We’re Here
To Help.
We’d love to connect. Follow us on social media or complete the form to ask a question or to be added to our company communications.


 Refer to the device User Manual for a list of contraindications, warnings, and cautions.
Refer to the device User Manual for a list of contraindications, warnings, and cautions. Training: Do not operate the Vanquish System without completing Francis Medical-provided physician training. Untrained operation of the device may lead to improper use. Improper use can result in patient injury or equipment malfunction.
Training: Do not operate the Vanquish System without completing Francis Medical-provided physician training. Untrained operation of the device may lead to improper use. Improper use can result in patient injury or equipment malfunction.