Press Release
Press Release: Francis Medical Adds Urology Company Coloplast to Its Roster of Investors
September 29, 2020
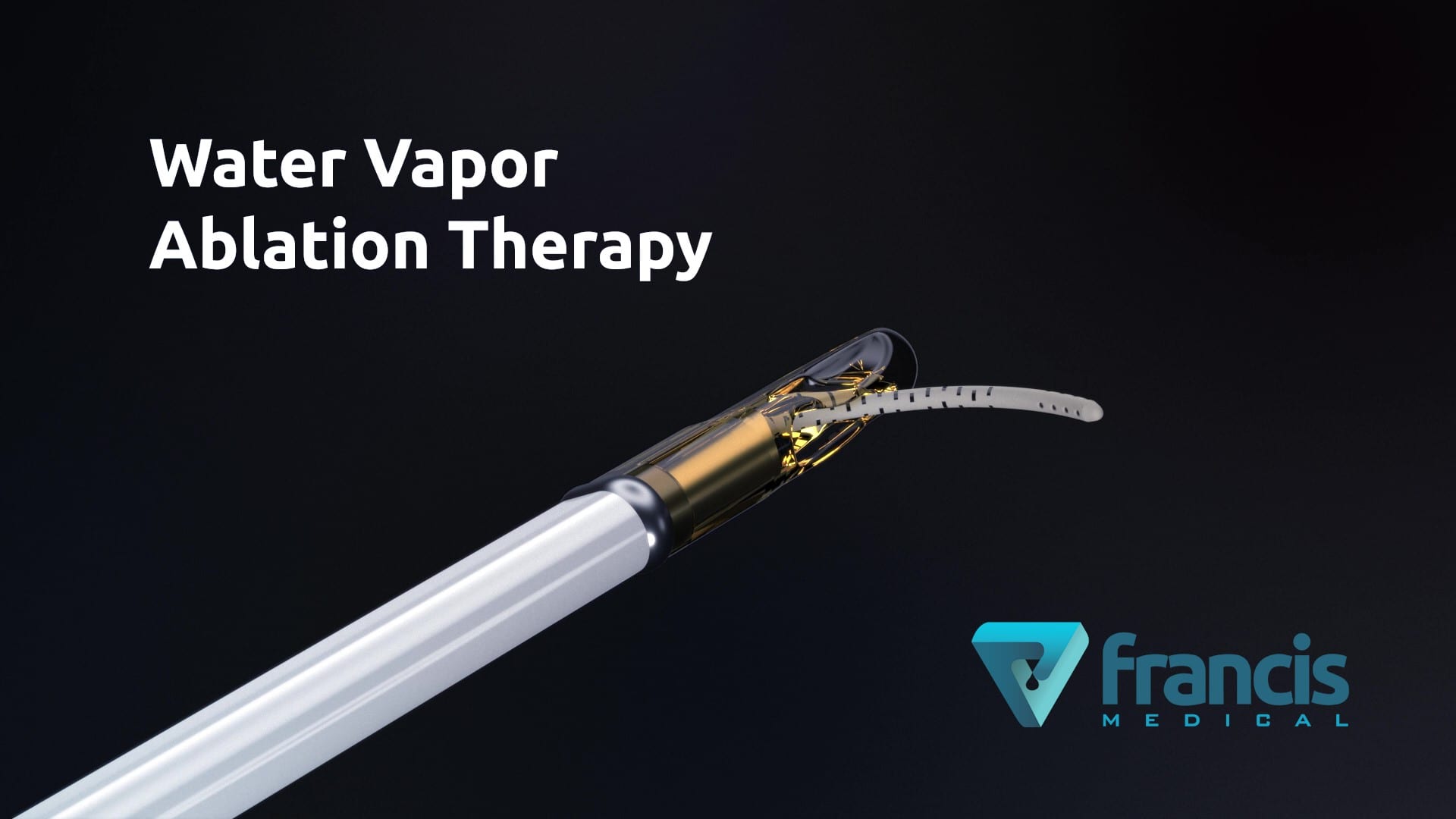
MINNEAPOLIS (Sept. 29, 2020) – Francis Medical, Inc., an early-stage company pioneering urological cancer treatments, beginning with prostate cancer, announced today that Coloplast SA has completed a $4 million equity investment in the company.
“We’re excited to bring on Coloplast, a strong company with deep experience in urology, as another strategic investor to help us continue developing urological cancer treatments that are tough on cancer, yet gentle on patients,” said Mike Kujak, Francis Medical President and CEO. “The investment also aligns well with the strategic direction of Coloplast’s Interventional Urology business, to bring innovative solutions to the urology market.”
Prostate cancer is the second most common cancer in American men. The American Cancer Society estimates that one in nine American men will be diagnosed with prostate cancer in their lifetime. It is a serious disease where current treatment options often cause further complications of urinary incontinence and erectile dysfunction. Francis Medical’s groundbreaking ablation technology applies the thermal energy stored in sterile water vapor to deliver targeted treatments to the cancerous tissue in a simple transurethral procedure, potentially minimizing the side effects often experienced with other prostate cancer treatments.
“Coloplast Interventional Urology is focused on bringing solutions to our existing customers in urology and gynecology,” said Steve Blum, Coloplast President of Interventional Urology. “Most of our existing urologist customers also see patients with prostate cancer, so we’re pleased to invest in a company dedicated to developing innovative and life-saving cancer treatments.”
Francis Medical’s VAPOR 1 U.S. early feasibility study is currently underway to evaluate the safety and efficacy of the company’s minimally invasive water vapor ablation therapy for the treatment of prostate cancer. Francis Medical received an investigational device exemption (IDE) for the study from the U.S. Food and Drug Administration (FDA) in late 2019. The outcomes of the VAPOR 1 study will be used to support a U.S. IDE submission to the FDA for the company’s pivotal trial. For more information on Francis Medical, visit www.francismedical.com.
About Francis Medical
Founded in 2018, Francis Medical is a privately held company committed to developing urological cancer treatments that are tough on cancer and gentle on patients, with a compassionate belief that cancer tissue can be treated with minimally invasive treatments. The foundation of the company is a tribute to and legacy of the inventor’s father, Francis Hoey, whose life was ended by prostate cancer with treatments that had harsh implications on his normal daily living. The platform technology utilizes water vapor energy to ablate targeted cancerous tissue while preserving surrounding structures, potentially minimizing the side effects often experienced with other cancer treatments. The company also has efforts underway in the areas of bladder and kidney cancer. For more information on Francis Medical, visit www.francismedical.com or call (763) 951-0370.
Press Release: Francis Medical Coloplast Investor Announcement
Recent Posts
University of Chicago Urologist Performs First Commercial Vanquish® Water Vapor Ablation Procedures in the State
websites2026-05-28T12:41:39-05:00May 28th, 2026|
Minneapolis [May 06, 2026] – Francis Medical, Inc., a privately held medical device company based in Maple Grove, Minn., today announced that Naveen Kella, MD, of The Urology Place has successfully completed the first commercial procedures in San Antonio, Texas, using the Vanquish® Water Vapor Ablation System, a minimally invasive technology designed to ablate targeted prostate tissue. Dr. Kella, who was also a clinical investigator in the VAPOR 2 pivotal study, performed the procedures on patients diagnosed with localized prostate cancer at Huebner Ambulatory Surgery Center.
Francis Medical Announces Further Expansion of Commercial Vanquish® Procedures to Ablate Prostate Tissue
websites2026-05-06T13:39:18-05:00May 6th, 2026|
Minneapolis [May 06, 2026] – Francis Medical, Inc., a privately held medical device company based in Maple Grove, Minn., today announced that Naveen Kella, MD, of The Urology Place has successfully completed the first commercial procedures in San Antonio, Texas, using the Vanquish® Water Vapor Ablation System, a minimally invasive technology designed to ablate targeted prostate tissue. Dr. Kella, who was also a clinical investigator in the VAPOR 2 pivotal study, performed the procedures on patients diagnosed with localized prostate cancer at Huebner Ambulatory Surgery Center.
Francis Medical Announces First Commercial Vanquish® Procedure to Ablate Prostate Tissue in the Midwest
websites2026-04-20T17:16:40-05:00April 21st, 2026|
Minneapolis [April 21, 2026] – Francis Medical, Inc., a privately held medical device company based out of Maple Grove, MN, today announced that Aaron Milbank, MD. of Minnesota Urology has successfully completed the first commercial procedure in the Midwest using the Vanquish® Water Vapor Ablation System, a minimally invasive technology designed to ablate targeted prostate tissue.





 Refer to the device User Manual for a list of contraindications, warnings, and cautions.
Refer to the device User Manual for a list of contraindications, warnings, and cautions. Training: Do not operate the Vanquish System without completing Francis Medical-provided physician training. Untrained operation of the device may lead to improper use. Improper use can result in patient injury or equipment malfunction.
Training: Do not operate the Vanquish System without completing Francis Medical-provided physician training. Untrained operation of the device may lead to improper use. Improper use can result in patient injury or equipment malfunction.